Polyethylene Glycol 400(for Injection)
(CDE REGISTRATION NO.: F20250000195)
Name: Polyethylene glycol 400 (for injection)
CAS No.: 25322-68-3
Other name: PEG-400
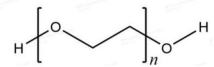
Molecular formula: H(OCH2CH2)nOH ,n=the average number of oxyvinyl groups.
Average molecular weight: 380-420
Quality Standard: USP-NF, EP, ChP2025, JP
Packaging: 0.5kg/bottle; 1kg/bottle/carton; 5kg/bottle/carton; ; 20kg/bottle/carton
Appearance: A colorless or nearly colorless viscous liquid
Solubility: Readily soluble in water or ethanol.
Structure:
Application:
| Application | Function | Advantage | Sample |
| Pharm grade | Solvent / Solubilizer | Good solubility and caneffectively dissolve a variety of drug components. | |
| Injection grade | Carrier and stabilizer Lyophilizationprotectant | As a carrier to facilitate drug absorption and delivery within the body.Enhancing the stability of proteins and peptides byforming a hydration layer Protecting drugs and aiding reconstitution in lyophilized injections | Azithromycin Injection |
| Injection grade | Plasticizer/Viscosity modifier | As a plasticizer, it improves flexibility and plasticity.Adjust viscosity to improve flowability and stability. |
Incompatibility
1. There is a risk of reaction with strong oxidizing agents, as the ether bonds and hydroxyl groups in its molecular structure readily react with oxidants.
2. It has a swelling or dissolving effect on plastics such as polyethylene, polyvinyl chloride, and phenolic resins, as well as cellulose ester filter membranes, which may affect the integrity of packaging containers or filtration systems.
Satety
Approved by the FDA for injectable use, it exhibits low toxicity and low immunogenicity, with no toxic reactions observed at final concentrations up to 50% (v/v) administered intravenously or intramuscularly.
Storage
1. Store in a cool, dry, and well-ventilated place, sealed, at an ambient temperature not exceeding 40°C. Avoid contact with humid air and oxidants.The prepared solution must be sealed and protected from light. It can be stored for 1 month at -20 °C and 6 months at -80°C. Repeated freeze-thaw cycles will cause the solution to lose its effectiveness; it is recommended to aliquot and use only as needed.
2. It contains a liquid crystalline phase and undergoes partial crystallization at extremely low temperatures (below 0 degrees Celsius).
